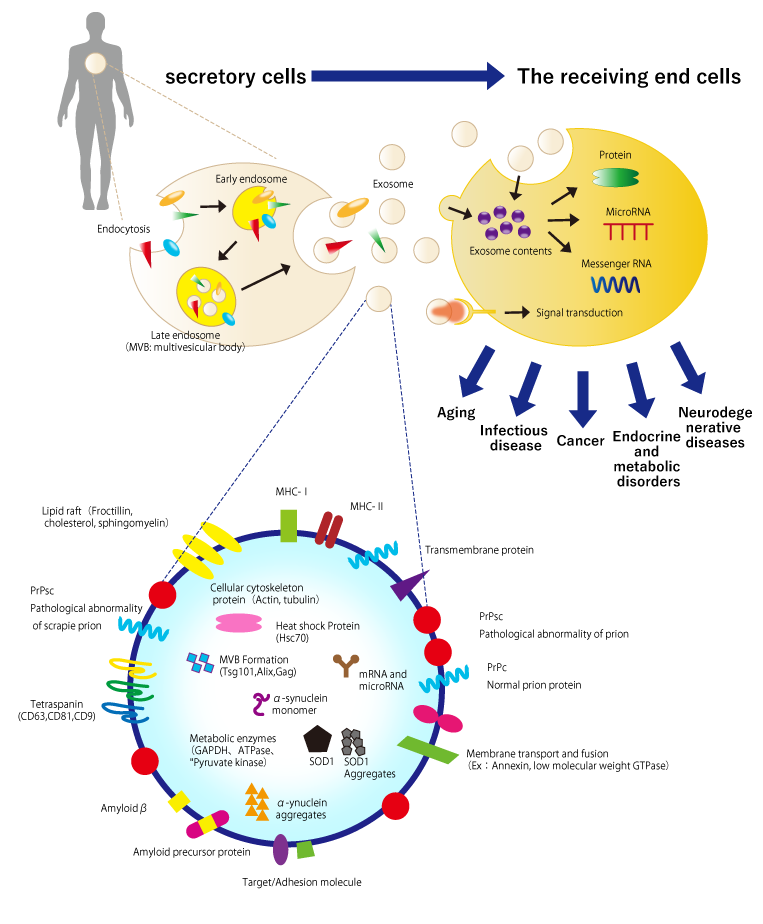
3. Maguire G. Stem cell therapy without the cells. Commun Integr Biol. 2013;6:e26631. DOI:10.4161/cib.26631.
5. Caplan AI, Correa D. The MSC: an injury drugstore. Cell Stem Cell. 2011;9:11-15.
13. Valadi H, Ekstrom K, Bossios A, et al. Exosome mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nature Cell Biol. 2007;9:654-659. Demonstration of exosome-mediated communication between cells during development
14. Ratajczak J, Miekus K, Kucia M, et al. Embryonic stem cell derived micro-vesicles reprogram hematopoietic pro-genitors: evidence for horizontal transfer of mRNA and protein delivery. Leukemia. 2006;20:847-856.
19. Yanez-Mo M, Siljander PR, Andreu Z, et al. Biological proper-ties of extracellular vesicles and their physiological functions. J Extracell Vesicles. 2015;4. DOI:10.3402/jev.v4.27066.
32. Deregibus MC, Cantaluppi V, Calogero R, et al. Endothelial progenitor cell derived microvesicles activate an angiogenic program in endothelial cells by a horizon-tal transfer of mRNA. Blood. 2007;110:2440-2448.
40. Teixeira FG, Carvalho MM, Sousa N, et al. Mesenchymal stem cells secretome: a new paradigm for central ner-vous system regeneration. Cell Mol Life Sci. 2013;70:3871-3882.
41. Vishnubhatla I, Corteling R, Stevanato L, et al. The devel-opment of stem cell-derived exosomes as a cell-free regenerative medicine. J Circ Biomark. 2014;3:2. DOI:10.5772/58597.
43. Liang X, Ding Y, Zhang Y, et al. Paracrine mechanisms of mesenchymal stem cell-based therapy: current status and perspectives. Cell Transplant. 2013;9:1045-1059.
44. van Koppen A, Joles JA, van Balkom BW, et al. Human embryonic mesenchymal stem cell-derived conditioned medium rescues renal function in rats with established chronic kidney disease. PLoS ONE.
(?)Ionescu L, Byrne RN, van Haaften T, et al. Stem cell conditioned medium improves acute lung injury in mice: in vivo evidence of stem cell paracrine action. Am J Physiol Lung Cell Mol Physiol. 2012;303:L967–97.
45. Ratajczak MZ, Kucia M, Jadczyk T, et al. Pivotal role of paracrine effects in stem cell therapies in regenerative medicine: can we translate stem cell-secreted paracrine factors and microvesicles into better therapeutic strate-gies. Leukemia. 2012;26:1166-1173.
50. Osugi M, Katagiri W, Yoshimi R, et al. Conditioned media from mesenchymal stem cells enhanced bone regenera-tion in rat calvarial bone defects. Tissue Eng Part A. 2012;18:1479-1489.
51. Maumus M, Jorgensen C, Noel D. Mesenchymal stem cells in regenerative medicine applied to rheumatic dis-eases: role of secretome and exosomes. Biochimie. 2013;95:2229-2234.
52. Toh WS, Foldager CB, Pei M, et al. Advances in mesench-ymal stem cell-based strategies for cartilage repair and regeneration. Stem Cell Reviews and Reports. 2014;10:686-696.
53. Akyurekli C, Le Y, Richardson RB, et al. A systematic review of preclinical studies on the therapeutic potential of mesenchymal stromal cell-derived microvesicles. Stem Cell Rev. 2015;11:150-160. Review of key preclinical studies associated with mesenchymal stem cell (MSC)-sourced exosomes
55. Gatti S, Bruno S, Deregibus MC, et al. Microvesicles derived from human adult mesenchymal stem cells pro-tect against ischaemia reperfusion induced acute and chronic kidney injury. Nephrol Dial Transplant. 2011;26:1474-1483.
56. Wang Y, Zhang L, Li Y, et al. Exosomes/microvesicles from induced pluripotent stem cells deliver cardioprotec-tive miRNAs and prevent cardiomyocyte apoptosis in the ischemic myocardium. Int J Cardiol. 2015;192:61-69.
57. Kilpinen L, Impola U, Sankkila L, et al. Extracellular membrane vesicles from umbilical cord blood derived MSC protect against ischemic acute kidney injury, a feature that is lost after inflammatory conditioning. J Extracell Vesicles. 2013;10:3402.
60. Lamichhane TN, Sokic S, Schardt JS, et al. Emerging roles for extracellular vesicles in tissue engineering and regen-erative medicine. Tissue Eng Part B Rev. 2015;21:45-54. DOI:10.1089/ten.teb.2014.0300.
61. Sahoo S, Klychko E, Thorne T, et al. Exosomes from human CD34+ stem cells mediate their proangiogenic paracrine activity. Circ Res. 2011;109:724-728.
62. Lee C, Mitsialis SA, Aslam M, et al. Exosomes mediate the cytoprotective action of mesenchymal stromal cells on hypoxia induced pulmonary hypertension. Circulation. 2012;126:2601-2611.
63. Zhang J, Guan J, Niu X, et al. Exosomes released from human induced pluripotent stem cells derived MSCs facilitate cutaneous wound healing by promoting col-lagen synthesis and angiogenesis. J Trans Med. 2015;13:49. DOI:10.1186/s12967-015-0417-0.
64. Ibrahim AG, Cheng K, Marban E. Exosomes as critical agents of cardiac regeneration triggered by cell therapy. Stem Cell Reports. 2014;2:606-619.
71. Lee HK, Finniss S, Cazacu S, et al. Mesenchymal stem cells deliver exogenous miRNAs to neural cells and induce their differentiation and glutamate transporter expres-sion. Stem Cells Dev. 2014;23:2851-2861.
73. Zhuang X, Xiang X, Grizzle W, et al. Treatment of brain inflammatory diseases by delivering exosome-encapsulated anti-inflammatory drugs from the nasal region to the brain. Mol Ther. 2011;19:1769-1779. DOI:10.1038/mt.2011.164.
74. Xin H, Li Y, Liu Z, et al. Systematic administration of exo-somes released from mesenchymal stromal cells promote functional recovery and neurovascular plasticity after stroke in rats. J Cereb Blood Flow Metab. 2013;33:1711-1715.
75. Zhang Y, Chopp M, Meng Y, et al. Effect of exosomes derived from multipluripotent mesenchymal stromal cells on functional recovery and neurovascular plasticity in rats after traumatic brain injury. J Neurosurg. 2015;122:856-867.
76. Doeppner TR, Herz J, Gorgens A, et al. Extracellular vesi-cles improve post-stroke neuroregeneration and prevent postischemic immunosuppression. Stem Cells Transl Med. 2015;4:1131-1143.
77. Xin H, Li Y, Liu Z, et al. MiR-133b promotes neural plas-ticity and functional recovery after treatment of stroke with multipotent mesenchymal stromal cells in rats via transfer of exosome-enriched extracellular particles. Stem Cells. 2013;31:2737-2746.
78. Bruce A, Ilagan R, Guthrie K, et al. Selected renal cells modulate disease progression in rodent models of chronic kidney disease via NFκB and TGFβ1 pathways. Regen Med. 2015;10:815-839.
79. Fleury A, Martinez MC, Le Lay S. Extracellular vesicles as therapeutic tools in cardiovascular diseases. Front Immunol. 2014;5:370. DOI:10.3389/fimmu.2014.00370.
82. Vlassov AV, Magdaleno S, Setterquist R, et al. Exosomes: current knowledge of their composition, biological func-tions, and diagnostic and therapeutic potentials. Biochimica et Biophysica Acta. 2012;1820:940-948.
83. Kordelas L, Rebmann V, Ludwig A-K, et al. 2013. MSC-derived exosomes: a novel tool to treat therapy-refractory graft-versus-host disease. Leukemia. 2014;28:970-973. DOI:10.1038/leu.2014.41.
101. Lee HJ, Lee EG, Kang S, et al. Efficacy of microneedling plus human stem cell conditioned medium for skin reju-venation: a randomized, controlled, blinded split face study. Ann Dermatol. 2014;26:584-591.
102. Harn HJ, Huang MH, Huang CT, et al. Rejuvenation of aged pig facial skin by transplanting allogeneic granulo-cyte colony stimulating factor induced peripheral blood stem cells from a young pig. Cell Transplant. 2013;22: 755-765.
103. Kim WS, Park BS, Park SH, et al. Antiwrinkle effect of adipose-derived stem cell: activation of dermal fibroblast by secretory factors. J Dermatol Sci. 2009;53:96-102.
104. Shim JH, Park JY, Lee MG, et al. Human dermal stem/ progenitor cell-derived conditioned medium ameliorates ultraviolet a induced damage of normal human dermal fibroblasts. PLoS One 2013;e67604. DOI:10.1371/journal. pone.0067604.
105. Chen CC, Murray PJ, Jiang TX, et al. Regenerative hair waves in aging mice and extra-follicular modulators Follistatin, Dkk1 and Sfrp4. J Invest Dermatol. 2014;134:2086-2096.
106. Fukuoka H, Suga H. Hair regeneration treatment using adipose-derived stem cell conditioned medium: follow-up with trichograms. Eplasty. 2015;15:e10.
107. Ahmed MI, Alam M, Emelianov VU, et al. MicroRNA-214 controls skin and hair follicle development by modulat-ing the activity of the Wnt pathway. J Cell Biol. 2014;207:549-567.
108. Zhang GE, Grizzle WE. Exosomes: a novel pathway of local and distant intercellular communication that facil-itates the growth and metastasis of neoplastic lesions. Am J Pathol. 2014;184:28-41.
109. Harris DA, Patel SH, Gucek M, et al. Exosomes released from breast cancer carcinomas stimulate cell movement. PLoS One 2015;e0117495. DOI:10.1371/journal.pone.0117495.
111. Li J, Sherman-Baust CA, Tsai-Turton M, et al. Claudin containing exosomes in the peripheral circulation of women with ovarian cancer. BMC Cancer. 2009;9:244. DOI:10.1186/1471-2407-9-244.
118. Mittelbrunn M, Gutierrez-Vazquez C, Villarroya-Beltri C, et al. Unidirectional transfer of microRNA-loaded exo-somes from T cells to antigen-presenting cells. Nature Commun. 2011;2:282. DOI:10.1038/ncomms1285.
119. www.systembio.com [cited 2015 Apr 11].
120. Basu J, Ludlow JW. MSC sourced exosomes as therapeu-tic agents for wound healing and skin regeneration: from scaled production to functional regenerative out-comes in vitro and in vivo. International Society for Stem Cell Research Annual Meeting; 2015 Jun 24-27; Stockholm.
121. Basu J, Ludlow JW. Developmental engineering the kid-ney: leveraging principles of morphogenesis for renal regeneration. Birth Defects Res C Embryo Today. 2012;96:30-38.
122. Genheimer CW, Ilagan RM, Spencer T, et al. Molecular characterization of the regenerative response induced by intrarenal transplantation of selected renal cells in a rodent model of chronic kidney disease. Cells Tissues Organs. 2012;196:374-384.
123. www.fda.gov/BiologicsBloodVaccines/default.htm [cited2015 Apr 11].